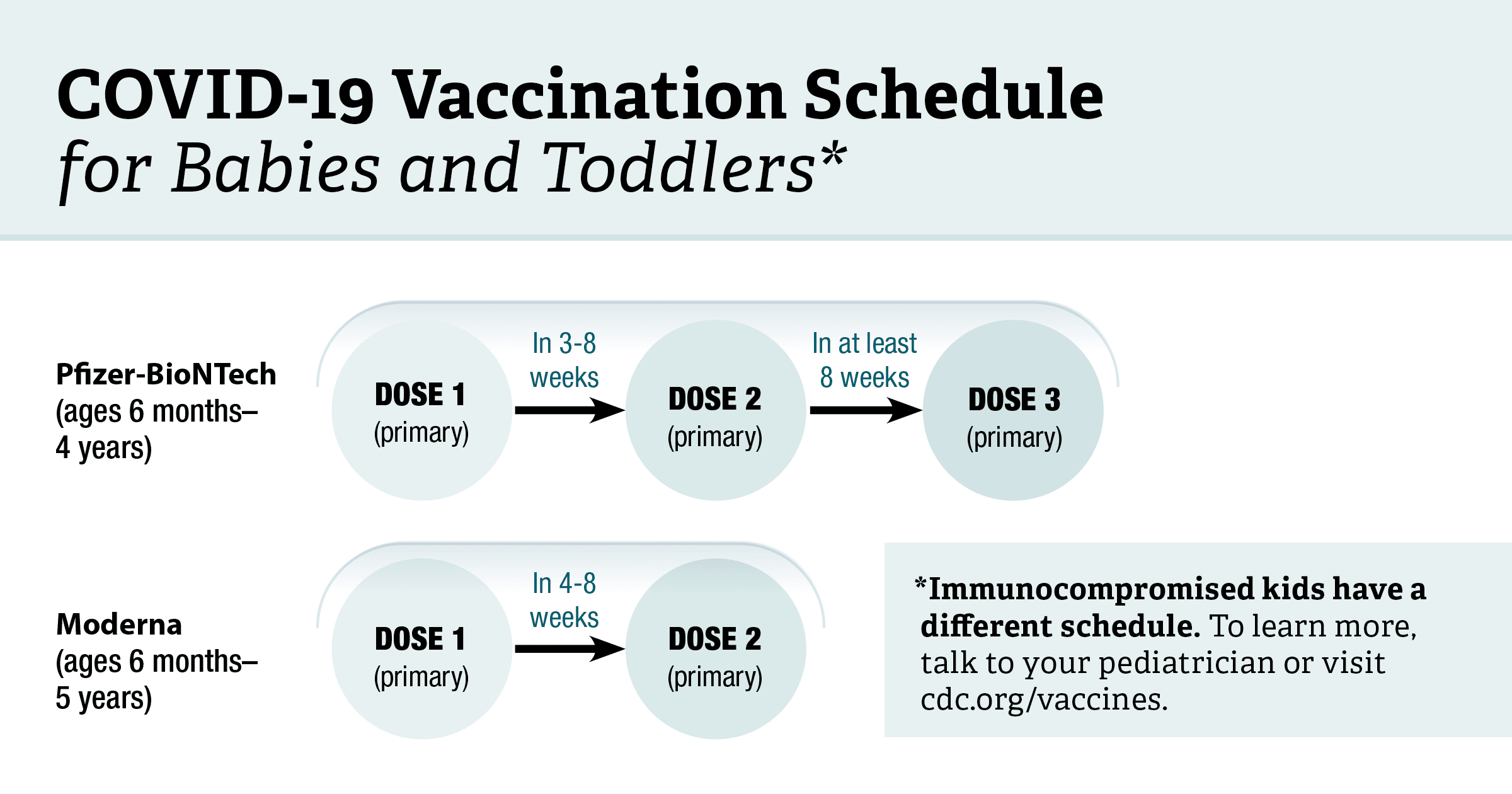
COVID-19 Vaccine Second-Dose Completion and Interval Between First and Second Doses Among Vaccinated Persons — United States, December 14, 2020−February 14, 2021 | MMWR
DPH Advises Vaccine Providers to Offer Pfizer COVID-19 Vaccine for Children 5 to 11 Years of Age - State of Delaware News

Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: an observational study - The Lancet

Syringe size and supply issues continue to waste COVID-19 vaccine doses in United States | Science | AAAS

3rd COVID-19 Vaccine Dose for Certain Immunocompromised Individuals - Anne Arundel County Department of Health
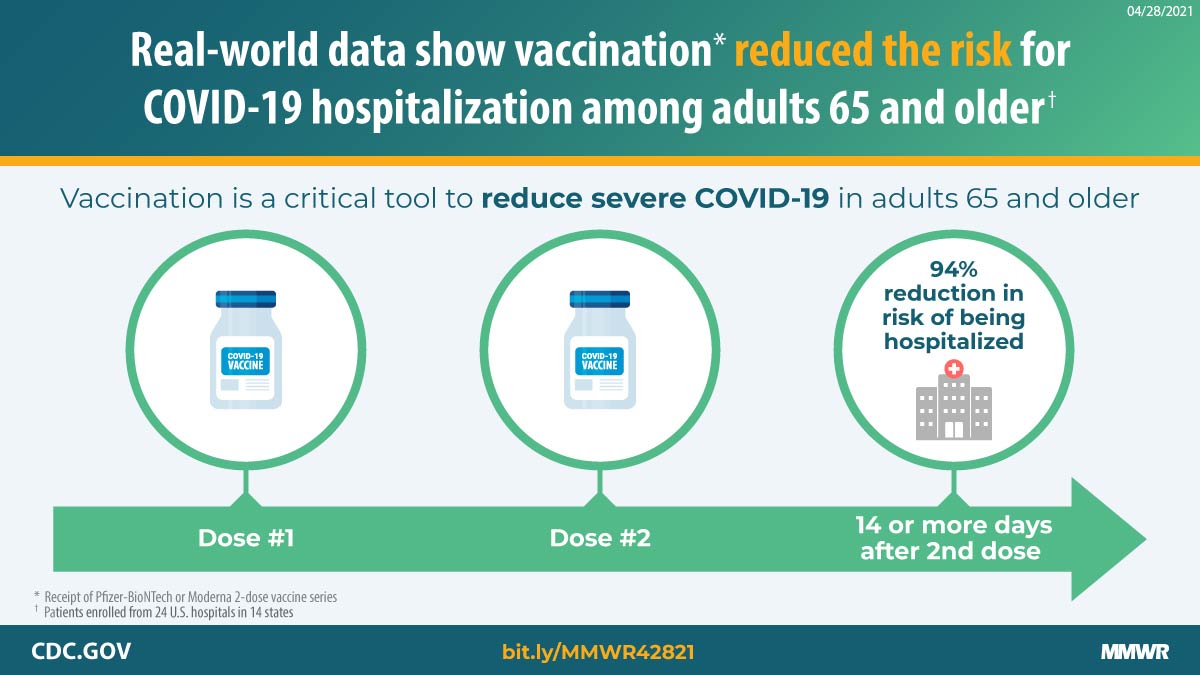
Effectiveness of Pfizer-BioNTech and Moderna Vaccines Against COVID-19 Among Hospitalized Adults Aged ≥65 Years — United States, January–March 2021 | MMWR
US CDC recommends third dose of Pfizer and Moderna COVID-19 vaccines for immunocompromised people | aidsmap
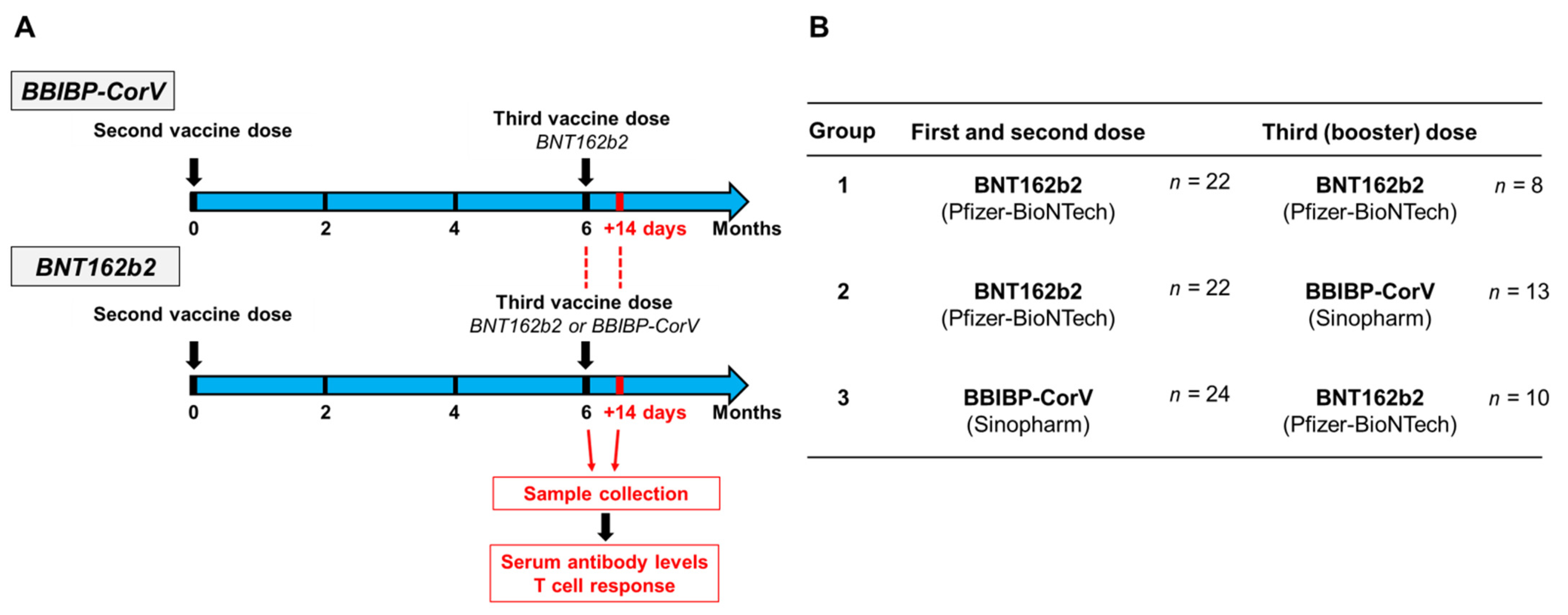
Vaccines | Free Full-Text | Antibody and T Cell Responses against SARS-CoV-2 Elicited by the Third Dose of BBIBP-CorV (Sinopharm) and BNT162b2 (Pfizer-BioNTech) Vaccines Using a Homologous or Heterologous Booster Vaccination Strategy